
How Health Economics Will Improve Clinical Trials to Maximize Health
Due to the increasing urgency of accessing healthcare services, and a patient-centric approach, decisions need to be more open, fair, and empowering of patient opinions. Health economics guarantees a decision-making framework.
“A healthy economy is a foundation for a healthy future.” (Charlie Kirk)
In response to the lack of resources, up-to-date professional education, and novel challenges, health economics is a novel field of study to support healthcare decisions while improving health outcomes. It provides a framework to facilitate decision-making and efficiency while positively impacting clinical practice.
What is health economics?
Health economics is an applied field of study systematically investigating various issues often encountered in healthcare. Hence, the medical system is explored through economic theories to understand the dynamic and decision-making process between patients, healthcare providers, public/private organizations, and governments. Therefore, health economics is implemented to promote health by applying theories of production, efficiency, competition, regulation, policies, evaluation of advanced technologies, investments, and strategic behavior.
What goods and services should we produce and how? Who should receive these services?
This discipline aims to answer simple questions by assuming the approach of generating and efficiency-maximizing benefits of resources.
Why should health economics be integrated into medical systems?
A few decades ago, any medical decisions were based on implicit values and determined exclusively by the physician. By contrast, due to the increasing urgency of accessing healthcare services, and a patient-centric approach, decisions need to be more open, fair and empowering of patient opinions. Therefore, health economics would guarantee a decision-making framework to organize operations addressing a wide range of healthcare challenges. Health economists are involved in the production of health operating at different levels from planning, budgeting, monitoring, and evaluating decisions to generate novel values and better healthcare delivery.
Furthermore, economic evaluation is central in this discipline because it aims at enhancing efficient choices in healthcare among different economic principles such as opportunity costs valuing the service and outcome, consumer/provider perspectives, and marginal analysis to estimate resources invested and benefits. Thus, the economic evaluation will measure and analyze the benefits of the:
- Cost minimization: analysis of costs between different interventions (generating the same consequences), where the most convenient intervention is selected.
- Cost-efficient: programs and treatments with the same health outcome are compared and the one with the most efficient results, based on a ratio scale, is selected.
- Cost-utility: analysis to assess costs and benefits of different interventions/programs to promote health longevity and life quality (e.g., when a new drug has higher costs and benefits over another, it should be calculated whether paying extra costs is worth the benefits gained).
- Cost-benefit: the analysis of costs and consequences of a treatment on a monetary basis. Hence, if the benefits are less than the costs, the treatment is considered convenient and accepted.
- Cost consequences: it is a different method to look at costs and benefits when making a decision. Thus, costs and outcomes are evaluated and presented separately while avoiding the decision-making processes to be affected by potential bias.
Health economics in clinical research
The demonstration that a new therapy is effective, is no longer sufficient to guarantee the best healthcare practice. Indeed, the increasing need for resources supporting care services, besides higher costs in developing novel treatments, constantly requires funds and finances. This process would influence those professionals in charge of deciding on whether a new treatment should be pursued even when available resources are limited. Therefore, an economic evaluation may be applied to estimate costs and benefits from the perspective of optimal clinical outcomes [1]. This fact is particularly true in the context of clinical trials in which a closer examination of the relationship between health outcomes and costs determines the viability of the study.
Accordingly, the result of this economic evaluation, based on a consistent set of data, would influence the choice of potentially adopting innovative technologies (e.g., diagnostic devices) based on the reimbursement costs, and thus an early estimation would be also necessary regarding the fulfillment of regulatory approvals. For this reason, to have better guidance in conducting proper economic evaluations in clinical trials, the book by Glick et al (2015) [2] on economic evaluation in clinical trials covers issues and techniques associated with the collection of costs and outcome data. Besides, it proposes a framework and a methodology (e.g., made of examples and exercises) for reporting and interpreting economic outputs related to clinical trials, thus supporting the reader in this learning process with applicable and practical solutions.
What are clinical trials?
Clinical research is medical research involving humans and it can be based on observational studies and clinical trials. Different from observational studies, which regard the recording of specific information (e.g., through medical exams, tests, questionnaires) about a group and observed over time to identify potential for clinical trials. Clinical trials are designed to gather data on the dosage, safety, and efficacy of new interventions/treatments while answering scientific questions about biomedical and behavioral research.
In general, clinical trials are the primary way to investigate whether a new treatment (e.g., a drug or medical device) is safer, more effective, and with lower side effects than other treatments. Other types of clinical trials are focused on prevention, thus aimed at detecting early-onset diseases, even before the symptoms appear. Nevertheless, before being tested in humans and getting approvals by the U.S. Food and Drug Administration (FDA) or after receiving health authority/ethics committee approval of the country where the trial is conducted, clinical trials are characterized by a large set of laboratory tests and studies on animal models to evaluate and determine how safe and effective the treatment under investigation is. After approval, the FDA allows the intervention to be tested in humans. Furthermore, in the process of establishing the feasibility and viability of a clinical trial, the evolution advances through four phases (and based on which the trials are defined) that test the potential of the treatment/intervention in terms of dosage, safety, and effectiveness, and side effects.
Phase I trial: it is the initial phase that tests an experimental treatment/intervention/device on a small sample of individuals (20 to 80), often healthy individuals, to evaluate its safety and slide effects to identify the proper dosage.
Phase II trial: continues the testing on a larger sample (100-300) to measure the effectiveness of the treatment/intervention/device. Thus, it aims at gathering data on whether the treatment under investigation works in individuals with a specific condition/disease.
Phase III trial: it aims to gain more information about the safety and effectiveness of the treatment/intervention/device on even larger samples (100-3000) while investigating different populations, dosages, and in combination with other medications. At this stage, FDA may approve or not the treatment or the device under experimentation.
Phase IV trial: it is the last phase of the cycle in which the treatment/intervention/device is monitored in a large and diverse population to evaluate its effectiveness and safety after the FDA approval. This is the stage in which potential side effects may emerge due to the greater sample size and a longer evaluation period.
Why participate in a clinical trial?
There might be several reasons why individuals participate in clinical trials. Most of the time, people join a clinical trial because the previous treatments were ineffective in improving their health. Thus, by participating in a clinical trial, they would have the opportunity to test innovative treatments before they are officially on the market. In addition, some study research is designed to involve healthy individuals but available to test new interventions aimed at preventing disease-onset (e.g., a particular disease that is common in their family). Another reason would be related to the need and interest in taking more action on health while supporting scientists in their research to improve health outcomes. Overall, independently of why people participate in clinical trials, those who join the process have the opportunity to be part of a potential and novel scientific discovery.
Moreover, clinical trials are designed to guarantee good scientific practice, validity, and reproducibility of outcomes. They may vary according to size and costs, conducted in single or multiple research institutes, in a single or different country. Lastly, due to their high costs, clinical trials are sponsored mainly by governments, organizations, pharma, biotech, or medical device companies.
A flexible approach to clinical trials
Limited financial resources affect healthcare decisions, mainly when investing in health research (e.g., new clinical trials and advanced technologies) [3]. Thus, a cost-effective evaluation should be considered to properly allocate these resources to improve health outcomes and patient care [4].
A Cost-effective analysis involves two relevant aspects of the health technology assessment (HTA). The first refers to the decision about whether it is cost-effective to invest in new technology while measuring the quality-adjusted life years (QALYs) of competing health technologies to compare their value and benefit to health based on their costs. The second decision regards whether it is cost-effective to invest and initiate a novel research study and measure based on the value of information analysis (VOIA) while assessing and collecting further information to conduct clinical trials.
Why adaptive clinical trials?
Usually, clinical trials are executed in three phases in which the trials are designed, conducted, and, after completion, acquired data are analyzed according to a specific methodology. Despite the linearity of this approach, it lacks flexibility without allowing any modification throughout the trial execution. Differently, adaptive designs (AD) were proposed as a flexible alternative in which pre-specified changes to the trial’s course can be applied concerning the evidence from data. These a priori planned adaptations are indeed different from unplanned ad hoc modifications (typically observed in traditional clinical trials), and therefore capable of maintaining the validity and integrity of the study. Pre-planned adaptations may relate to sample size re-definition, discard of specific treatments/doses, allocation ratio of patients to specific groups, and early-stage trial suspension (e.g., successful or not effective). Furthermore, in ADs, data are constantly collected, analyzed, and examined to ensure good clinical practice and that the trial is structured in response to a specific research question. The flexibility gathered through ADs is also a better condition for patients, who would benefit the most compared to traditional designs. However, although ADs have been made available for several years, they are still not well established and implemented in the clinical practice due to a lack of experience, strict regulations, or lack of clarity regarding how AD should be properly carried out [5].
Hence, a tutorial paper published in BMC Medicine (2018) [5] aimed to encourage the implementation of ADs in clinical trials by providing guidance and references for establishing flexible designs. Indeed, AD would allow better use of resources (e.g., time and money), resulting in more efficient, ethical, and informative designs than traditional fixed designs. In addition, the authors highlighted pivotal issues and challenges often encountered in these designs regarding funding, ethical approvals, communication with stakeholders, and trial implementation with participants while giving a few suggestions about how to overcome these obstacles. Lastly, transparency and reproducibility, central principles in clinical practice, were also examined with a focus on how data are interpreted and reported.
Health economics in adaptive clinical trials
As previously discussed, to accomplish a reliable and effective health economic analysis, adaptive designs are alternative, flexible, and innovative methods to execute clinical trials. Different from traditional designs based on fixed sample sizes, in adaptive designs data, are examined in parallel with the progression and results of the trial to gather insight into the viability of the study. Hence, time and resources can be better estimated for the experimenter and for the patients [6,7] to demonstrate their clinical effectiveness. Nevertheless, a closer assessment regarding the impact that health economic analysis has on adaptive designs is still unclear, and which opportunities may bring needs more investigation.
Accordingly, a recent study published in the journal Trials (2020) [3] explored the perspective of focus groups (e.g., stakeholders, researchers, decision-makers, general population) based on semi-structured interviews on using health economics to enhance the effectiveness of adaptive clinical trials. The qualitative study implemented a pragmatic approach in which a thematic framework (based on literature research and discussion with advisory groups) and a framework investigating three main areas in healthcare (e.g., ethical, methodological, and practical issues) were adopted to identify themes in the transcripts. Results demonstrated that the focus should be on answering clinical research questions, despite the relevance of cost-effectiveness for decision-making. While analyzing and designing adaptive clinical trials, health economics has the potential to enhance the efficiency of health technologies, although a few recommendations were proposed to properly take advantage of health economic principles in establishing novel methods in clinical trials. These recommendations were defined from the themes collected in the data and corresponding to different views of different stakeholders regarding the HTA process, experience from research teams, and the public advisory group.
A few recommendations were:
- early examination of the trial data to update the health economic model and that these data are properly collected through the trial progression;
- use a hierarchy of rules to make decisions based on cost-effectiveness according to clinical outcomes;
- use health economic data to modify the trial progression;
- adoption of an innovative mindset to welcome and recognize the role of health economics and cost-effectiveness in decision-making;
- develop software, and case studies to support researchers in understanding the method while learning to integrate this approach into their method;
- searching for different funding sources to foster novel designs and methodologies;
- evaluation of additional costs for implementing (non-)adaptive clinical trials;
- shared expertise within the research community to improve trial outcomes and economic analysis.
In several countries, decision-makers rely on scientific and social values when allocating resources, in which a cost-effective evaluation is required before initiating any medical intervention. Accordingly, differences between clinical and economic evaluation would emerge concerning the purpose of the research and the methodology adopted to foster the investigation. A potential mismatch may arise because evidence of clinical benefit is not always a prerequisite for economic analysis (e.g., large studies demonstrate benefits but costly, small clinical studies are cost-effective but less reliable), and where a more compatible approach between clinical and economic evaluation is needed [8]. Hence, these recommendations were proposed to ensure clinical effectiveness and that proper training and resources are allocated to promote the realization of this approach [3] while considering the clinical relevance (e.g., patient-level benefit) as a value around which economic analysis is made.
Economic evaluation and clinical trials
Safety, effectiveness, and efficacy are the main factors based on which clinical trials are designed and in which a decision is made combining technological advances, costs, and patient benefits. Hence, typical decisions are made based on whether a new technology or treatment should be introduced, whether a specific patient group would benefit from it, whether a specific technology or intervention is better than another, and who is in charge of funding those costs. These questions are crucial in healthcare and anytime a decision is made explicitly, another decision is made implicitly. Indeed, the decision to invest in a novel technology, considered to be effective and safe in clinical practice, translates into the question “where should the resources be allocated?.” Thus, the novel technology acquires a monetary value that is evaluated.
What is an economic evaluation?
In this framework, the economic evaluation requires a fair judgment to promote a novel set of values in healthcare and specific criteria were also proposed to suggest how this analysis should be conducted and which might be potential implications for medical research [9]. The economic evaluation is used to inform the medical system while making decisions and policies. In particular, considering the low availability of resources in health systems, informed and supervised decisions would generate a greater impact on the clinical trial outcome.
Therefore, an economic evaluation is a formal and systematic comparison of the costs and benefits associated with two or more interventions. Hence, it compares the additional net costs and net health benefits between an existing treatment and a novel one. By measuring costs and outcomes, it is possible to compute an estimation of the average costs and benefits across patients and controls (groups) participating in the treatment. Therefore, a cost-effectiveness estimation of the novel treatment (over the existing treatment) can be drawn according to the costs and outcome measures.
The advantage of conducting economic analysis in clinical trials was already advanced by a research of 2005 published in Value Health journal [10] which highlighted blinding and randomization as strengths, followed by the potential of collecting patient data (costs and outcomes) alongside the trial execution, making it less costly than a “stand-alone” economic evaluation. However, potential issues were discussed by a research published in Health Economics the year after. Hence, more attention should be paid to follow-up that is usually too short for adequate economic analysis, the trial setting (healthcare systems across different countries may vary thus influencing decisions), and the observation and evaluation of other trials' outcomes and epidemiological data. Therefore, a successful economic evaluation in a clinical trial requires close collaboration between health economists and clinical researchers across the stages process [12] ensuring that health economists participate in the trial design phases to properly plan and allocate resources. Nevertheless, there is not a “gold standard” rule of what defines a suitable economic study, and different guidelines on methods for economic evaluations were proposed by different organizations [13]. Although the BCL of economic quality [14] is the most used, others are the Consensus Health Economic Criteria [15] Quality of Health Economic Studies [16], and the Drummond checklist [17].
From a practical perspective, an economics evaluation compares the gained benefits of investing in a new intervention over a standard care procedure based on its costs. Thus, the results of this analysis can be visualized graphically in which the relation between incremental costs (y-axis) and effects (x-axis) is shown, for which a decision can be made. Based on this cost-effectiveness plane each condition can be evaluated, and in which an incremental cost-effectiveness ratio (ICER) is also estimated (green dash line) indicating that all points on or under this line can be considered cost-effective [13].
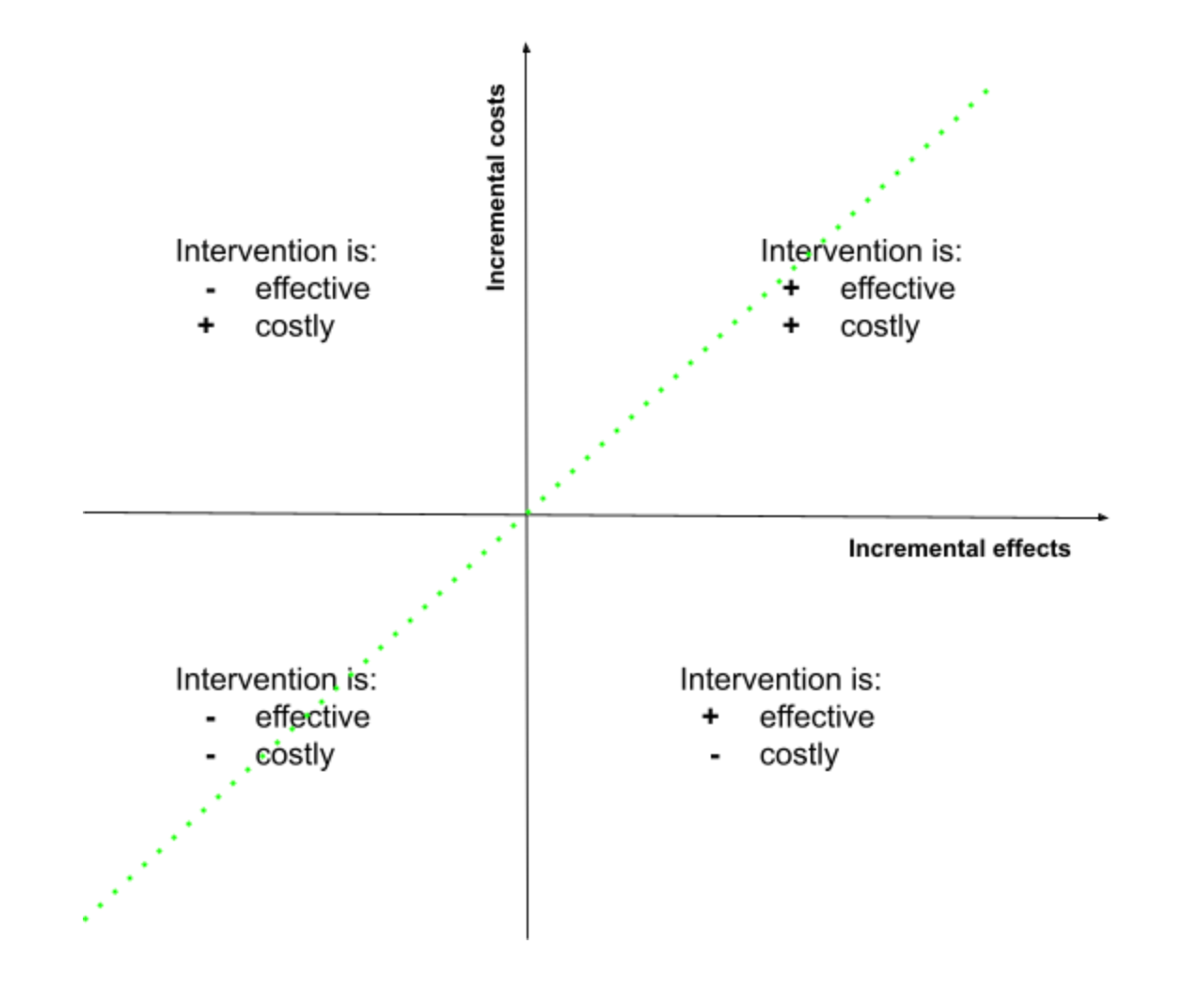
Several types of economic evaluation can be differentiated based on the approach implemented to measure the outcome. For example, the cost-benefit analysis considers benefits in terms of monetary value. Cost-utility analysis (CUA) relies on a single measure that combines the length of life and its quality, often called quality-adjusted life-years (QALYs). Cost-effectiveness analysis uses a measure of health (e.g., life-years saved) to estimate the outcome. Finally, the cost-minimization analysis identifies similar interventions based on their effectiveness but differs by their costs (to be compared). In addition, other analyses can be defined as cost-only which do not represent a typical economic evaluation but may help gather information for decision-making regarding the intervention [13].
Moreover, consideration should be made in the conclusion that a clinical study might differ from an economic study. Being the ICER, a relevant measure in an economic evaluation, in which cost and health economic outcomes are highlighted and that is different from clinical outcomes alone. In addition, whether an economic evaluation would rely on a QALY measure, a clinical trial would primarily evaluate the clinical outcome. Even statistical analysis methods may differ. Indeed, a clinical trial would rely on the statistical significance that the procedure is effective, rather than the probability that the intervention is cost-effective as the economic evaluation would do. Nevertheless, to better investigate how close an economic and clinical evaluation is, the Health Technology Assessment (HTA) program would be useful [13]. For more information about the economic analysis alongside clinical trials read here.
References:
- Manns BJ. The role of health economics within clinical research. Methods Mol Biol. 2009;473:235-50. doi: 10.1007/978-1-59745-385-1_14. PMID: 19160742.
- Glick, H. A., Doshi, J. A., Sonnad, S. S., & Polsky, D. (2015). Economic Evaluation in Clinical Trials. (Second Edition). Oxford university press.
- Flight, L., Julious, S., Brennan, A. et al. How can health economics be used in the design and analysis of adaptive clinical trials? A qualitative analysis. Trials 21, 252 (2020). https://doi.org/10.1186/s13063-020-4137-2
- Drummond MF, Sculpher MJ, Claxton K, Stoddart GL, Torrance GW. Methods for the economic evaluation of health care programmes. Oxford: Oxford University Press; 2015.
- Pallmann, P., Bedding, A.W., Choodari-Oskooei, B. et al. Adaptive designs in clinical trials: why use them, and how to run and report them. BMC Med 16, 29 (2018). https://doi.org/10.1186/s12916-018-1017-7
- Bauer P, Brannath W. The advantages and disadvantages of adaptive designs for clinical trials. Drug Discov Today. 2004; 9(8):351–7.
- Chow S, Chang M. Adaptive design methods in clinical trials - a review. Orphanet J Rare Dis. 2008; 3(11):11.
- Whitehurst, D.G., Bryan, S. Trial-based clinical and economic analyses: the unhelpful quest for conformity. Trials 14, 421 (2013). https://doi.org/10.1186/1745-6215-14-421
- Michael F. Drummond, Greg L. Stoddart, Economic analysis and clinical trials, Controlled Clinical Trials, Volume 5, Issue 2, 1984, Pages 115-128, ISSN 0197-2456. https://doi.org/10.1016/0197-2456(84)90118-1
- O’Sullivan AK, Hompson D, Rummond MF. Collection of health economic data alongside clinical trials: is there a future for piggyback evaluations? Value Health 2005;8:67–79. 10.1111/j.1524-4733.2005.03065.x
- Sculpher M, Claxton K, Drummond M. Whither trial-based economic evaluation for health care decision making? Health Econ 2006;15:677–87. 10.1002/hec.1093.
- Petrou S, Gray A. Economic evaluation alongside randomised controlled trials: design, conduct, analysis, and reporting. BMJ 2011;342:14. 10.1136/bmj.d1548
- Raftery J, Young A, Stanton L, Milne R, Cook A, Turner D, Davidson P. Clinical trial metadata: defining and extracting metadata on the design, conduct, results and costs of 125 randomised clinical trials funded by the National Institute for Health Research Health Technology Assessment programme. Health Technol Assess. 2015 Feb;19(11):1-138. doi: 10.3310/hta19110. PMID: 25671821; PMCID: PMC4781629.
- Drummond MF, Jefferson TO. Guidelines for authors and peer reviewers of economic submissions to the BMJ. The BMJ Economic Evaluation Working Party. BMJ1996;313:275–83. 10.1136/bmj.313.7052.275.
- Evers S, Goossens M, de Vet H, van Tulder M, Ament A. Criteria list for assessment of methodological quality of economic evaluations: consensus on health economic criteria. Int J Technol Assess Health Care 2005;21:240–5.
- Chiou CF, Hay JW, Wallace JF, Bloom BS, Neumann PJ, Sullivan SD, et al. Development and validation of a grading system for the quality of cost-effectiveness studies. Med Care2003;41:32–44. 10.1097/00005650-200301000-00007
- Anon. Critical Assessment of Economic Evaluation. In Drummond MF, Sculpher MJ, Torrance GW, O’Brien BJ, Stoddart GL, editors. Methods for the Economic Evaluation of Health Care Programmes. Oxford: Oxford University Press; 2005. pp. 27–54.

ExO Insight Newsletter
Join the newsletter to receive the latest updates in your inbox.









